Atlanta, Georgia — Verint Systems Inc. (“VSI”) of Melville, New York and its wholly owned subsidiary Verint Americas Inc. (“VAI”) of Alpharetta, Georgia (collectively, “Verint”) have sued Interactive Intelligence, Inc. of Indianapolis, Indiana in the Northern District of Georgia for infringement of twenty separate patents; these patents have been registered with the U.S. Patent Office.
Verint is engaged in the business of inventing, developing, manufacturing, selling, installing, and/or distributing computer software and hardware products and systems. The products and systems are directed to, inter alia, the analysis, recording, monitoring, transmission, and/or security of electronic communications, such as telephonic, facsimile, and e-mail communications to and from contact centers and call centers which handle incoming and/or outgoing contacts with actual and prospective customers and clients. Verint’s technology can provide an end user with the ability to capture, analyze and act on large volumes of complex information sources, such as voice, video, and unstructured text, which can enhance the ability of organizations of all sizes to make better decisions based on such information. Verint’s main location for research and development relating to recording technology is located at VAI’s facility in Georgia.
Verint’s products and systems are used by more than 10,000 organizations in over 150 countries, including over 80 percent of the Fortune 100. For example, Verint’s workforce-optimization and voice-of-the-customer solutions are designed to help organizations enhance customer-service operations in contact centers, branches, and back-office environments. This can increase customer satisfaction, reduce operating costs, uncover revenue opportunities and improve profitability. Verint uses its core competencies to develop highly scalable solutions with advanced, integrated analytics for both unstructured and structured information. Verint asserts that it has expended substantial resources inventing and developing this technology. Verint has licensed one or more of its patents to others in the industry through its Open Innovation Network (“OIN”) licensing program.
Defendant Interactive, a competitor of Verint, is in a similar business. It manufactures, uses, sells, offers to sell, installs, distributes, exports, and/or imports computer software and hardware products and systems directed to and for use in connection with methods involving the analysis, recording, monitoring, transmission and security of electronic communications, such as telephonic communications to and from contact centers and call centers.
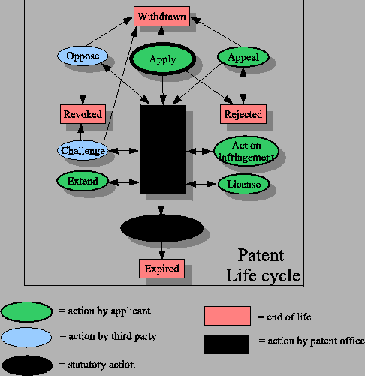
Verint contends that it is the sole owner of all rights to the patents at issue in this suit: U.S. Patent No. 5,790,798 (“the ‘798 patent”); U.S. Patent No. 7,203,285 (“the ‘285 patent”); U.S. Patent No. 7,376,735 (“the ‘735 patent”); U.S. Patent No. 7,574,000 (“the ‘000 patent”); U.S. Patent No. 7,774,854 (“the ‘854 patent”); U.S. Patent No. 7,852,994 (“the ‘994 patent”); U.S. Patent No. 7,903,568 (“the ‘568 patent”); U.S. Patent No. 8,204,056 (“the ‘056 patent”); U.S. Patent No. 8,285,833 (“the ‘833 patent”); U.S. Patent No. RE41,534 (“the ‘534 patent”); U.S. Patent No. RE43,324 (“the ‘324 patent”); U.S. Patent No. 8,401,155 (“the ‘155 patent”); U.S. Patent No. 8,359,434 (“the ‘434 patent”); U.S. Patent No. 8,345,828 (“the ‘828 patent”); U.S. Patent No. 8,275,944 (“the ‘944 patent”); U.S. Patent No. 8,204,053 (“the ‘053 patent”); U.S. Patent No. RE43,386 (“the ‘386 patent”); U.S. Patent No. 8,130,926 (“the ‘926 patent”); U.S. Patent No. 6,404,857 (“the ‘857 patent”) and U.S. Patent No. 6,510,220 (“the ‘220 patent”) (collectively, “the Patents-in-Suit”).
Verint believes that Interactive is, in various ways, infringing upon the Patents-In-Suit. In a letter to Interactive dated September 13, 2010, VSI invited Defendant to participate in its OIN. The OIN is a licensing program under which VSI offers to grant a worldwide license to its portfolio of patents directed, in part, to the analysis, recording, monitoring, transmission, and/or security of electronic communications. In its September 2010 letter, VSI provided Interactive with claim charts showing how Interactive’s activities fell within the claim scope of various patents within the OIN. Over the course of several months, several more letters were sent by VSI to Interactive, inviting it to participate in the OIN. No agreement was reached. The current lawsuit was subsequently filed by Verint.
Defendant Interactive is accused of infringing the Patents-in-Suit in violation of 35 U.S.C. § 271, including as follows:
(a) by making, using, offering to sell, and/or selling in the United States or importing into the United States computer software and/or hardware and/or systems, and/or by engaging in or practicing in the United States methods or processes covered by the Patents-in-Suit, including such methods or processes which utilize one or more of the Accused Products [those products of Interactive which are claimed to infringe on Verint’s patents]; and/or
(b) by offering to sell or selling within the United States or importing into the United States a component of a product or system within one or more of the patents of the Patents-in-Suit, or a material or apparatus for use in practicing a method or process within one or more of the patents of the Patents-in-Suit, constituting a material part of one or more of the patents of the Patents-In-Suit, knowing the same to be especially made or especially adapted for use in an infringement of one or more of the patents of the Patents-in-Suit, and not a staple article or commodity of commerce suitable for substantial non-infringing use; and/or
(c) by supplying or causing to be supplied in or from the United States all or a substantial portion of components to form a product or system within one or more of the patents in the Patents-in-Suit, including by supplying or causing to be supplied in or from the United States the Accused Products, in such a manner as to actively induce the combination of such components outside of the United States in a manner that would infringe one or more of the patents of the Patents-in-Suit if such combination occurred within the United States; and/or
(d) by supplying or causing to be supplied in or from the United States a component of a product or system within one or more of the patents of the Patents-in-Suit that is especially made or especially adapted for use according to one or more of the patents of the Patents-in-Suit and is not a staple article or commodity of commerce suitable for substantial non-infringing use, knowing that such component is so made or adapted and intending that such component will be combined outside of the United States in a manner that would infringe one or more of the patents of the Patents-in-Suit if such combination occurred within the United States, including by supplying or causing to be supplied in or from the United States one or more of the Accused Products; and/or
(e) actively inducing one or more of the activities identified in subparagraphs (a) through (d).
Interactive is accused of infringing, contributing to the infringement of, and actively inducing infringement of the Patents-in-Suit. The complaint lists twenty counts of patent infringement:
- Count I: Infringement of the ‘798 patent
- Count II: Infringement of the ‘285 patent
- Count III: Infringement of the ‘735 patent
- Count IV: Infringement of the ‘000 patent
- Count V: Infringement of the ‘854 patent)
- Count VI: Infringement of the ‘994 patent
- Count VII: Infringement of the ‘568 patent
- Count VIII: Infringement of the ‘056 patent
- Count IX: Infringement of the ‘833 patent
- Count X: Infringement of the ‘534 patent
- Count XI: Infringement of the ‘324 patent
- Count XII: Infringement of the ‘155 patent
- Count XIII: Infringement of the ‘434 patent
- Count XIV: Infringement of the ‘828 patent
- Count XV: Infringement of the ‘944 patent
- Count XVI: Infringement of the ‘053 patent
- Count XVII: Infringement of the ‘386 patent
- Count XVIII: Infringement of the ‘926 patent
- Count XIX: Infringement of the ‘857 patent
- Count XX: Infringement of the ‘220 patent
In the complaint, patent attorneys for Verint assert that Interactive had actual knowledge and/or notice from a time prior to the filing of the present complaint of at least the following: the ‘798 patent; the ‘285 patent; the ‘735 patent; the ‘000 patent; the ‘854 patent; the ‘994 patent and the ‘534 patent. It is also alleged that Interactive’s acts of infringement have been carried out deliberately and willfully, without the consent of Verint, at least with respect to the ‘798 patent; ‘285 patent; the ‘735 patent; the ‘000 patent; the ‘854 patent; the ‘994 patent and the ‘534 patent, entitling Verint to treble damages under 35 U.S.C. § 284. Verint further contends that this is an exceptional case entitling Verint to an award of attorneys’ fees under 35 U.S.C. § 285.
Plaintiff Verint asks for a judgment that Defendant Interactive has been and is infringing, is contributing to the infringement of, and is actively inducing infringement of the Patents-in-Suit; preliminary and permanent injunctive relief against such infringement under 35 U.S.C. § 283; damages, including treble damages, by reasons of Defendant’s acts of purportedly deliberate and willful infringement; attorneys’ fees; and costs, including expert witness fees.
Practice Tip: Verint is no stranger to the value of intellectual property. It utilizes more than 1,000 employees and contractors in research and development throughout the world, and has obtained or filed more than 570 patents and applications worldwide. In its last fiscal last year, Verint obtained issuance or allowance of 60 patents and applications in the United States.
Verint is also no stranger to intellectual property lawsuits. In 2008, a Georgia jury found that NICE Systems Ltd. had infringed Verint’s U.S. Patent No. 6,404,857 and awarded Verint $3.3 million in damages. A week later, in a separate lawsuit, Verint was found not to have infringed NICE’s U.S. Patent No. 6,871,229. There have been at least three other patent infringement lawsuits between Verint and NICE. There were rumors in January that NICE was negotiating to acquire Verint Systems, Inc.
Continue reading
 Washington, D.C. — The Federal Circuit ruled on two patent infringement decisions, Los Angeles Biomedical Research Institute v. Eli Lilly & Co. and Eli Lilly & Co. v. Los Angeles Biomedical Research Institute, that involve Indianapolis-based Eli Lilly and Company.
Washington, D.C. — The Federal Circuit ruled on two patent infringement decisions, Los Angeles Biomedical Research Institute v. Eli Lilly & Co. and Eli Lilly & Co. v. Los Angeles Biomedical Research Institute, that involve Indianapolis-based Eli Lilly and Company. Indiana Intellectual Property Law News
Indiana Intellectual Property Law News