 Cheng USA, Inc., a company based in Elkhart, Indiana and doing business under the names Arterra Distribution and WFCO Technologies, has filed a lawsuit in the United States District Court for the Northern District of Indiana against Kunshan Jinhui New Energy Technology Co., Ltd., which operates under the brand name Bestrenogy. The defendant is based in Jiangsu, China.
Cheng USA, Inc., a company based in Elkhart, Indiana and doing business under the names Arterra Distribution and WFCO Technologies, has filed a lawsuit in the United States District Court for the Northern District of Indiana against Kunshan Jinhui New Energy Technology Co., Ltd., which operates under the brand name Bestrenogy. The defendant is based in Jiangsu, China.
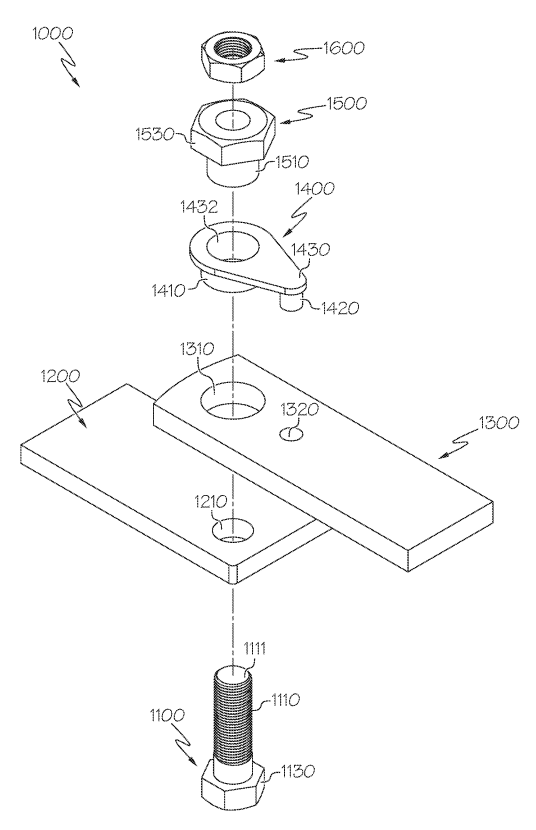
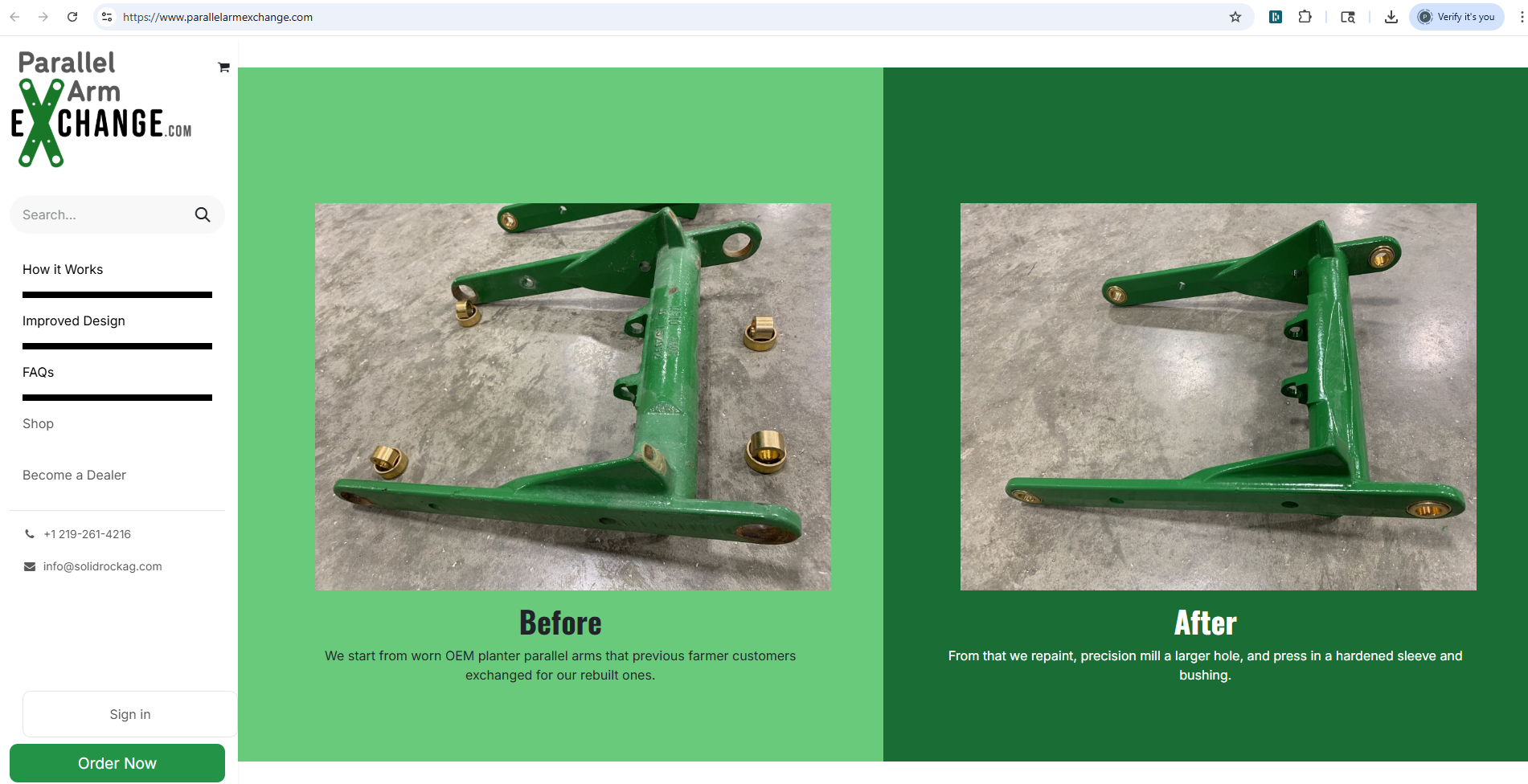
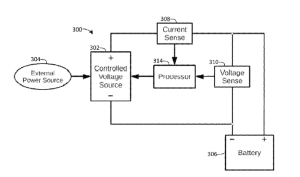
The lawsuit alleges that Bestrenogy is infringing on Cheng’s U.S. Patent No. 12,113,377, which covers a specific type of power conversion technology used in their popular WF-9855 converter charger. According to Cheng, Bestrenogy has been manufacturing, importing, using, and selling — or offering to sell — its AD-CB Battery Charger 9800 Series in the United States, and that this product incorporates technology covered by Cheng’s patent without authorization. The complaint includes images and descriptions of Bestrenogy’s product sold on Amazon.com that Cheng claims directly infringes on the patent.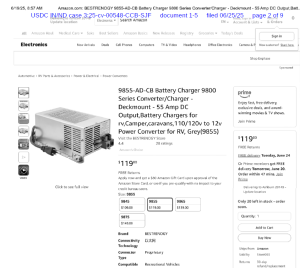
Cheng further accuses Bestrenogy of willful infringement, meaning it believes the defendant knew about the patent and still proceeded with the infringing activity. In addition to patent infringement, Cheng is also bringing claims of unfair competition and false designation of origin. The company alleges that Bestrenogy’s marketing materials, including the use of certain symbols, customer service contacts, and product descriptions, could mislead consumers into believing Bestrenogy’s products are associated with or approved by Cheng.
 Indiana Intellectual Property Law News
Indiana Intellectual Property Law News